
If no family member is a suitable donor, a matched unrelated donor can be used. Stanford doctors can now deplete T cells that cause graft-versus-host disease from the stem cell graft before transplantation, reducing the risk of GVHD and enabling every patient to find a donor. In the past, using a mismatched donor could increase the chances of developing graft-versus-host disease (GVHD).

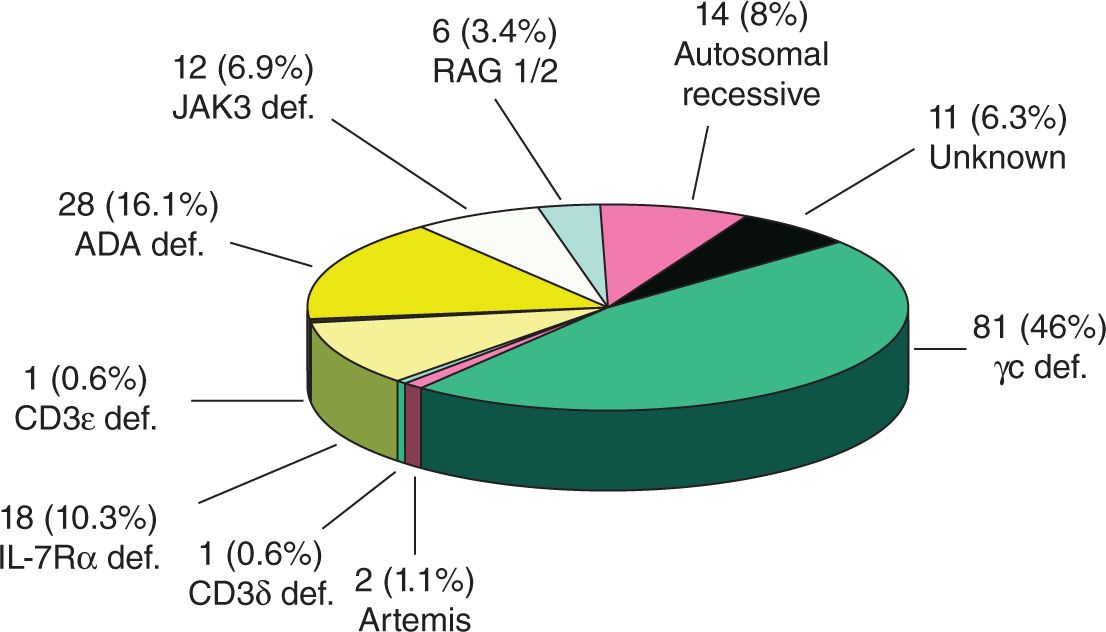
If a healthy matched sibling donor is not available, then stem cell transplantation from a half-matched, or haploidentical, family donor, usually the mother, is the next-best option. The therapy of choice for most patients with SCID is stem cell transplantation from a healthy matched donor, usually a sibling. The only curative therapy for SCID is stem cell transplantation from a healthy individual or with the patient’s own genetically modified cells, also known as gene therapy. How is SCID treated at Stanford Medicine Children’s Health? Your child’s diagnosis will be confirmed by genetic testing and an assessment of your child’s T and B cell numbers and function. Patients with SCID are categorized by their primary genetic defect, but common to all forms of SCID is an absence of protective T and B cells (immune cells that help attack invaders like bacteria, viruses, and fungi). Now, your child may have been diagnosed based on a newborn screening before he or she developed any infections. In the past, patients were diagnosed based on their clinical presentation or family history. Symptoms include diarrhea, fungal infections, and poor physical growth. What are the symptoms of SCID, and how is it diagnosed? A variety of genetic defects can produce SCID. Patients with severe combined immunodeficiency (SCID) are born without a functioning immune system and therefore are at increased risk of infections and death before the age of 2. 8.What is severe combined immunodeficiency (SCID)? Newborn screening for severe combined immunodeficiency in 11 screening programs in the United States. Kwan A, Abraham RS, Currier R, Brower A, Andruszewski K, Abbott JK, Baker M, Ballow M, Bartoshesky LE, Bonilla FA, Brokopp C, Brooks E, Caggana M, Celestin J, Church JA, Comeau AM, Connelly JA, Cowan MJ, Cunningham-Rundles C, Dasu T, Dave N, De La Morena MT, Duffner U, Fong CT, Forbes L, Freedenberg D, Gelfand EW, Hale JE, Hanson IC, Hay BN, Hu D, Infante A, Johnson D, Kapoor N, Kay DM, Kohn DB, Lee R, Lehman H, Lin Z, Lorey F, Abdel-Mageed A, Manning A, McGhee S, Moore TB, Naides SJ, Notarangelo LD, Orange JS, Pai SY, Porteus M, Rodriguez R, Romberg N, Routes J, Ruehle M, Rubenstein A, Saavedra-Matiz CA, Scott G, Scott PM, Secord E, Seroogy C, Shearer WT, Siegel S, Silvers SK, Stiehm ER, Sugerman RW, Sullivan JL, Tanksley S, Tierce ML, Verbsky J, Vogel B, Walker R, Walkovich K, Walter JE, Wasserman RL, Watson MS, Weinberg GA, Weiner LB, Wood H, Yates AB, Puck JM, Bonagura VR. Autosomal, sporadic, or the X-linked form may affect the neonate, and without treatment, patients rarely survive beyond one year of age before succumbing to opportunistic infections. Also, these infections may lead to early death in severe combined immunodeficiency disease, differentiating this condition from other forms or combined immunodeficiency.īoth T and B cell functions are disturbed or absent entirely in severe combined immunodeficiency disease. The onset of the clinical manifestations occurs by 6 months of age or before, with bacterial, viral, fungal and protozoal infections. Severe combined immunodeficiency disease (SCID) is the most severe expression among the combined immunodeficiency disorders. Immunotherapy sometimes is not available to treat these recurrent infections. These patients are susceptible to infection by many organisms. Patients with combined immunodeficiency disorder (T and B lymphocyte deficiency) present with recurrent infections usually early in life.
